1. Saponification – It is the alkaline hydrolysis of fats and oil.
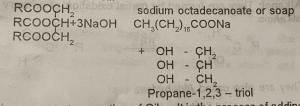
2. Hydrogenation of Oils – It is the process of adding hydrogen to the oils so as to harden them. It occurs in the presence of finely divided nickel.
1. Saponification – It is the alkaline hydrolysis of fats and oil.

2. Hydrogenation of Oils – It is the process of adding hydrogen to the oils so as to harden them. It occurs in the presence of finely divided nickel.